Does Gold Conduct Electricity
Gold is a unique metal known for its inertness and conductivity. While gold is an excellent conductor of electricity, its ability to conduct is not as high as that of other metals like copper or silver. In its elemental form, gold readily conducts electricity due to its free electrons, allowing for the flow of electric current. The conductivity of gold is often utilized in various electronic applications, such as connectors and switches. However, the use of a "chemistry balancing equations calculator" is unrelated to gold's conductivity. Balancing equations is a fundamental aspect of chemistry that ensures conservation of mass and charge in chemical reactions. It involves adjusting coefficients to achieve equality between the number of atoms on both sides of the equation. This process is crucial for understanding and predicting chemical reactions, but it does not directly relate to the electrical conductivity of gold.
Can Gold Conduct Electricity?
Gold is an excellent conductor of electricity, a property that contributes to its widespread use in various electronic applications. Unlike other metals, gold's conductivity is not affected by tarnish or corrosion, making it highly reliable for electrical connections. Gold's ability to conduct electricity is attributed to its atomic structure, where its outer electrons are loosely bound and can move freely. This free movement of electrons allows for efficient flow of electrical current.
Due to its conductivity and resistance to corrosion, gold is often employed in the manufacturing of connectors, switches, and other components in electronic devices. The use of gold in the production of high-quality electrical contacts ensures stable and low-resistance connections. While gold is a valuable precious metal, its role in the field of electronics highlights its practical applications beyond its aesthetic and monetary value. In summary, gold's exceptional ability to conduct electricity makes it a preferred material in the realm of electronics, contributing to the reliability and efficiency of various technological devices.
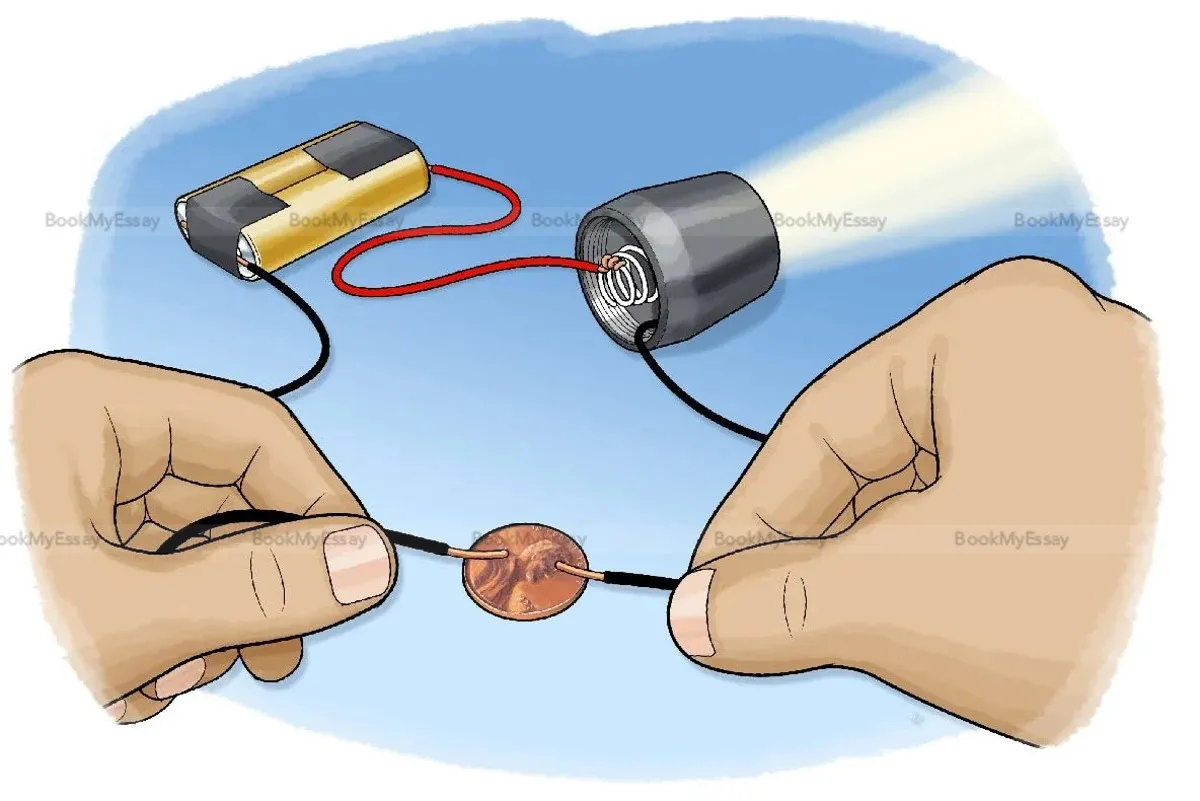
Is Gold An Electrical Conductor?
Seeking assignment help online to understand the conductivity of gold in electrical circuits is a wise decision. Gold, a noble metal, is indeed an excellent electrical conductor. Its conductivity arises from the fact that gold atoms have a relatively high number of free electrons, allowing for the easy flow of electric current. This property makes gold particularly valuable in various technological applications, such as electronics and telecommunications.
When students engage in online assignment help, they can explore the unique characteristics of gold as an electrical conductor. The assistance provided by online platforms often includes explanations of the underlying principles, helping learners grasp the concepts more effectively. Additionally, students can gain insights into practical applications of gold's conductivity, such as its use in connectors and components where reliable and efficient electrical conductivity is crucial.
In conclusion, utilizing online assignment help resources can enhance one's understanding of gold as an electrical conductor, unraveling its significance in modern technology and facilitating academic success in related studies.
Does Gold Possess Electrical Conductivity?
Why does Metal Conduct Electricity?
Gold is a metal renowned for its lustrous appearance and rarity, but does it possess electrical conductivity? The answer is yes. In the realm of metals, electrical conductivity is a characteristic linked to the presence of free electrons. Metals have a unique atomic structure that allows electrons to move freely, creating a flow of electric current when a voltage is applied. Gold, despite being a noble metal, retains this property due to its atomic structure. The outer electron shell of gold atoms contains free electrons that can easily move through the lattice structure of the metal.
The key lies in the sea of delocalized electrons shared among gold atoms. This mobility of electrons enables gold to conduct electricity efficiently. While gold is not as conductive as some other metals like copper or silver, its conductivity, coupled with its resistance to corrosion and tarnish, makes it valuable in various electronic applications, including connectors and coatings for sensitive electronic components. Understanding the relationship between metal conductivity and electron mobility sheds light on gold's role in the electrical conductivity spectrum.
Is Gold A Good Conductor Of Electricity?
When evaluating gold's conductivity, it's essential to recognize its unique properties. While gold is an exceptional conductor of electricity, its reputation as a premium material for electrical conductivity stems from its low resistivity and oxidation resistance. Gold boasts a conductivity rating surpassed only by silver and copper, making it a valuable choice in various electronic applications. The metal's conductivity arises from its atomic structure, allowing electrons to move freely. This feature is crucial for minimizing energy loss in electronic circuits. Gold's resistance to corrosion ensures long-term reliability in electrical connections. As an assignment help provider, understanding the distinctive characteristics of gold in conductivity becomes imperative. Students benefit from comprehending how gold's electrical properties contribute to its widespread use in electronics, from connectors to delicate circuitry. In conclusion, gold undeniably stands as a stellar conductor of electricity, solidifying its indispensable role in the realm of electrical engineering and design.
Is There Any Content On BookMyEssay That Explains The Electrical Conductivity Of Gold?
BookMyEssay is an academic writing service that offers assistance on various topics, including electrical conductivity and material properties. If you're looking for information on the electrical conductivity of gold, it's advisable to check their website or contact their customer support for the latest offerings.
Gold is renowned for its exceptional electrical conductivity, making it a valuable material in various industries, particularly in electronics. Its ability to efficiently conduct electricity is attributed to its unique atomic structure, which allows electrons to move freely. Understanding the electrical properties of gold is crucial in applications like circuitry and technology. BookMyEssay may provide insightful content on this subject, offering explanations, analyses, and academic support for individuals seeking a comprehensive understanding of the electrical conductivity of gold.






 3 Bellbridge Dr, Hoppers Crossing, Melbourne VIC 3029
3 Bellbridge Dr, Hoppers Crossing, Melbourne VIC 3029




