Metal That Conducts Electricity
Metal that conducts electricity refers to any metallic substance capable of allowing the flow of electrical current through it. This property is crucial in various applications, including electrical wiring, circuitry, and electronic devices. Metals conduct electricity due to the presence of free electrons within their atomic structure. These free electrons are able to move freely throughout the metal lattice, carrying electrical charge with them.
Common examples of metals that conduct electricity include copper, aluminum, silver, gold, and iron. Copper, in particular, is widely used in electrical wiring and electronics due to its excellent conductivity and relatively low cost. Aluminum is another commonly used metal in electrical applications, prized for its lightweight and corrosion resistance.
Metals conduct electricity efficiently because they have a high density of free electrons and a crystalline structure that allows for easy electron movement. Additionally, metals tend to have low resistance, meaning they offer little opposition to the flow of electrical current. This makes them ideal materials for conducting electricity in various industries and technologies.
Which Metal Is The Best Conductor Of Electricity?
The ability of metals to conduct electricity is a fundamental property that plays a crucial role in various technological applications. Among metals, silver is widely regarded as the best conductor of electricity. It boasts high electrical conductivity due to its unique atomic structure, allowing electrons to move freely through the lattice.
Gold is another metal renowned for its conductivity, albeit not as efficient as silver. Gold's conductivity is influenced by its resistance to corrosion and tarnish, making it a preferred choice for electronic components that require reliability over time.
While silver and gold are exceptional conductors, copper is the most commonly used metal in electrical applications due to its high conductivity, affordability, and abundance. Aluminum is also a good conductor, widely used in power transmission lines.
While gold does conduct electricity, it is not the most efficient choice. Silver holds the title of the best conductor, followed closely by copper, making them indispensable in the realm of electrical engineering and technology.
How Does The Conductivity Of Copper Compare To Other Metals?
When delving into the realm of materials science, understanding the conductivity of metals is essential. Copper, renowned for its exceptional electrical conductivity, stands out among its metallic counterparts. Its conductivity is second only to silver, making it a popular choice in electrical applications. Copper's high conductivity is attributed to its atomic structure, allowing electrons to move freely through its lattice.
Comparing copper to other metals, it surpasses most in terms of electrical conductivity. Aluminum, although widely used in electrical applications, exhibits lower conductivity than copper. Gold and silver boast higher conductivity than copper, but their prohibitive cost limits their widespread use in practical applications.
In academic writing, students often explore the electrical properties of metals, necessitating a comprehensive understanding of copper's conductivity compared to other metals. When seeking assignment help online, students can access valuable resources and guidance to enhance their knowledge of these topics. The conductivity of metals is a critical aspect of electrical engineering and physics, making it an intriguing subject for academic exploration.
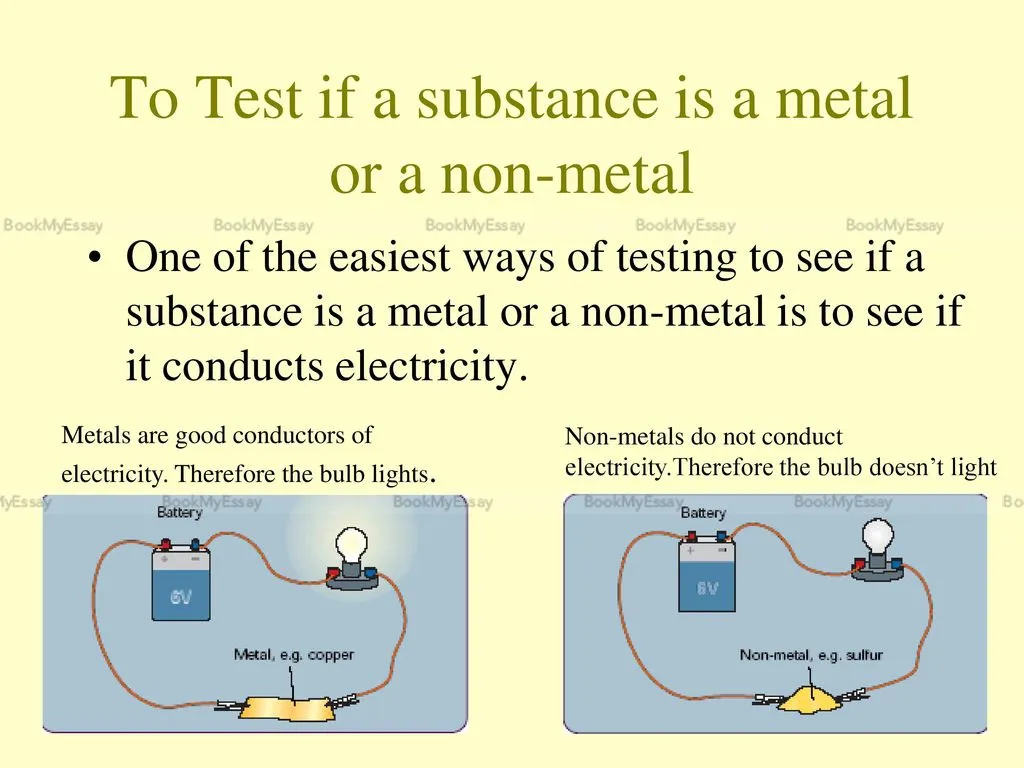
Can You List Three Common Metals That Conduct Electricity?
Assignment Assistance is crucial for students seeking guidance on various topics. In the realm of science and physics, understanding materials that conduct electricity is fundamental. Three common metals renowned for their electrical conductivity are copper, aluminum, and silver.
Copper is widely used in electrical wiring due to its exceptional conductivity, low resistance, and affordability. It is a preferred choice in various electrical applications, from power lines to electronic devices. Aluminum, although not as conductive as copper, is still a common choice for electrical transmission lines due to its lighter weight and cost-effectiveness. Silver, being one of the best conductors of electricity, is employed in specialized applications where maximum conductivity is imperative, like high-frequency circuits.
Students seeking Assignment Assistance often explore the properties and applications of these metals in electrical systems. Understanding the conductivity of different materials is essential in designing efficient and effective electrical components, making it a crucial aspect of various science and engineering assignments. Assignment Provider play a significant role in aiding students to grasp and apply these concepts effectively.
Features of Using BookMyEssay to Order Assignment Solutions
BookMyEssay offers a plethora of features that make it an ideal platform for ordering assignment solutions. Firstly, the website boasts a team of highly qualified experts across various subjects and disciplines, ensuring that students receive high-quality and well-researched content tailored to their specific requirements. Additionally, the platform offers 24/7 customer support, allowing students to seek assistance and clarifications at any time.
Another key feature of BookMyEssay is its commitment to confidentiality and originality. All assignments are thoroughly checked for plagiarism and confidentiality is maintained throughout the process, ensuring that students can submit their work with confidence. Furthermore, the platform offers competitive pricing and flexible payment options, making it accessible to students with varying budgets.
Overall, BookMyEssay provides a convenient and reliable solution for students seeking assistance with their assignments. With its team of experts, round-the-clock support, and commitment to quality and confidentiality, it stands out as a trusted platform for academic support.
As for the question "Why does metal conduct electricity?", it's because metals have free electrons that can move easily between atoms when a voltage is applied, creating an electric current. This is due to the metallic bonding in which positively charged metal ions are held together by a "sea" of delocalized electrons, allowing them to flow freely and conduct electricity.







 3 Bellbridge Dr, Hoppers Crossing, Melbourne VIC 3029
3 Bellbridge Dr, Hoppers Crossing, Melbourne VIC 3029




